Contents
1. Application type
The variable to be predicted can have two values (malignant or benign tumor). Therefore, this is a binary classification project.
The goal is to model the probability of a malignant tumor conditioned on the fine needle aspiration (FNA) test features using artificial intelligence and machine learning.
2. Data set
Data source
The breast_cancer.csv file contains the data for this example. Target variables can only have two values in a classification model: 0 (false) or 1 (true). The number of instances (rows) in the data set is 683, and the number of variables (columns) is 10.
Variables
The number of input variables, or attributes for each sample, is 9. All input variables are numeric-valued and represent measurements from digitized histopathological images of a fine-needle aspiration (FNA) biopsy. The target variable is 1, diagnose (0 or 1) benign (non-cancerous) or malignant (cancerous), respectively. The following list summarizes the variables information:
- clump_thickness: (1-10). Benign cells tend to be grouped in monolayers, while cancerous cells are often grouped in multilayers.
- cell_size_uniformity: (1-10). Cancer cells tend to vary in size and shape.
- cell_shape_uniformity: (1-10). Cancer cells tend to vary in shape and size.
- marginal_adhesion: (1-10). Normal cells tend to stick together, while cancer cells tend to lose this ability, so the loss of adhesion is a sign of malignancy.
- single_epithelial_cell_size: (1-10). It is related to the uniformity mentioned above. Epithelial cells that are significantly enlarged may be malignant.
- bare_nuclei: (1-10). This term is used for nuclei not surrounded by cytoplasm (the rest of the cell). Those are typically seen in benign tumors.
- bland_chromatin: (1-10). Describes a uniform “texture” of the nucleus seen in benign cells. In cancer cells, the chromatin tends to be more coarse and to form clumps.
- normal_nucleoli: (1-10). Nucleoli are small structures seen in the nucleus. In normal cells, the nucleolus is usually very small, if visible. The nucleoli become more prominent in cancer cells, and sometimes there are multiple.
- mitoses: (1-10). Cancer is essentially a disease of uncontrolled mitosis.
- diagnose: (0 or 1). Benign (non-cancerous) or malignant (cancerous) lump in a breast.
Instances
Finally, the use of all instances is set. Note that each instance contains a different patient’s input and target variables. The data set is divided into training, validation, and testing subsets. Neural Designer automatically assigns 60% of the instances for training, 20% for generalization, and 20% for testing. The user can choose to modify these values to the desired ones.
Variables statistics
Once we have set the data, we can perform a few related analytics. We check the provided information and ensure that the data is quality.
We can calculate the data statistics and draw a table with descriptive statistics (minimums, maximums, means, and standard deviations) of all variables in the data set. The next table depicts the values.
| Minimum | Maximum | Mean | Deviation | |
|---|---|---|---|---|
| clump_thickness | 1 | 10 | 4.44 | 2.82 |
| cell_size_uniformity | 1 | 10 | 3.15 | 3.07 |
| cell_shape_uniformity | 1 | 10 | 3.22 | 2.99 |
| marginal_adhesion | 1 | 10 | 2.83 | 2.86 |
| single_epithelial_cell_size | 1 | 10 | 3.23 | 2.22 |
| bare_nuclei | 1 | 10 | 3.54 | 3.64 |
| bland_chromatin | 1 | 10 | 3.45 | 2.45 |
| normal_nucleoli | 1 | 10 | 2.87 | 3.05 |
| mitoses | 1 | 10 | 1.6 | 1.73 |
| diagnose | 0 | 1 | 0.35 | 0.447 |
All variables are ranked from 1 to 10, with one meaning that the variable for that sample is low and ten meaning that it is high, e.g., a value of clump_thickness equal to 1 would mean that this sample clump thickness is low. As we can see in the previous table, the mean for all the variables is less than 5. The input variable with the minor standard deviation is “mitoses”.
Variables distributions
Also, we can calculate the distributions for all variables. The following figure shows a pie chart with the numbers of malignant (positive) and benign (negative) tumors in the data set.
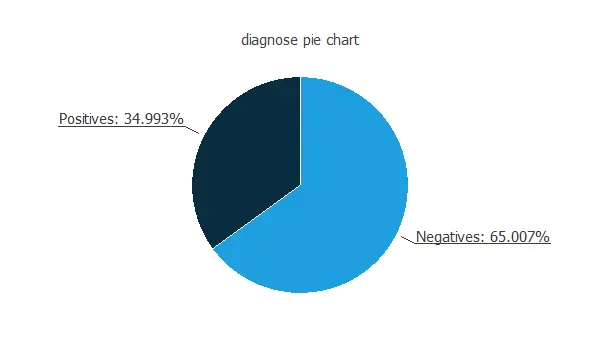
As depicted in the image, malignant tumors represent 35% of the samples, and benign tumors represent approximately 65%.
Inputs-targets correlations
The inputs-targets correlations might indicate which factors most influence whether a tumor is malignant or benign and, therefore, be more relevant to our analysis.
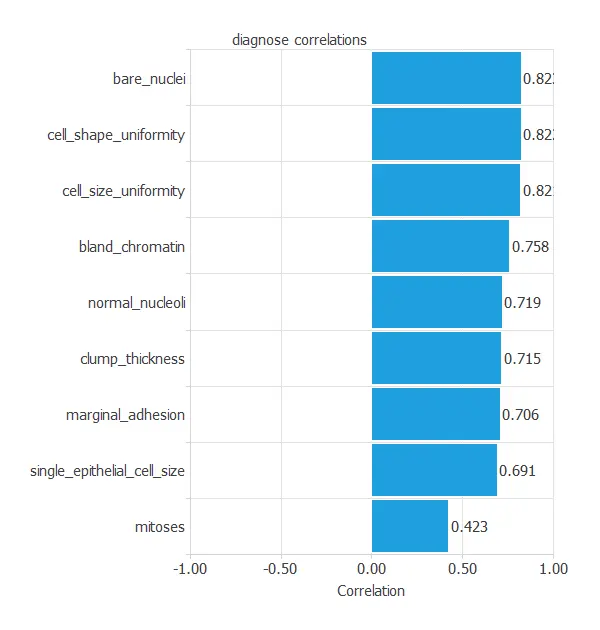
Here, the most correlated variables with malignant tumors are bare nuclei, cell shape uniformity, and cell size uniformity.
3. Neural network
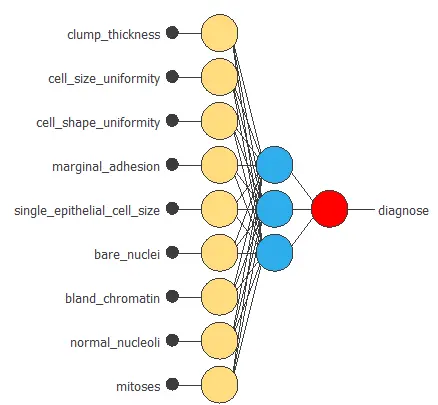
The next step is to set up a neural network to represent the classification function. For this class of applications, the neural network is composed of:
The scaling layer contains the statistics on the inputs calculated from the data file and the method for scaling the input variables. Here, we have set the minimum-maximum method. Nevertheless, the mean-standard deviation method would produce very similar results. The scaling layer has nine inputs since there are nine input variables.
A perceptron layer with a hidden logistic layer. The neural network must have nine inputs since the number of scaling neurons is nine. As an initial guess, we use three neurons in the hidden layer.
The probabilistic layer only contains the method for interpreting the outputs as probabilities. Indeed, as the sum of all outputs from a probabilistic layer must be 1, two methods would always yield one here since there is only one output. Moreover, as the output layer’s activation function is logistic, that output can already be interpreted as a probability of class membership.
The probabilistic layer has three inputs. It also has one output, representing the probability of a sample being a malignant tumor.
The following figure is a graphical representation of this neural network for breast cancer diagnosis.
4. Training strategy
The fourth step is to set the training strategy, which is composed of two terms:
- A loss index.
- An optimization algorithm.
The loss index is the weighted squared error with L2 regularization. This is the default loss index for binary classification applications.
We can state the learning problem as finding a neural network that minimizes the loss index. That is a neural network that fits the data set (error term) and does not oscillate (regularization term).
The optimization algorithm that we use is the quasi-Newton method. This is also the standard optimization algorithm for this type of problem.
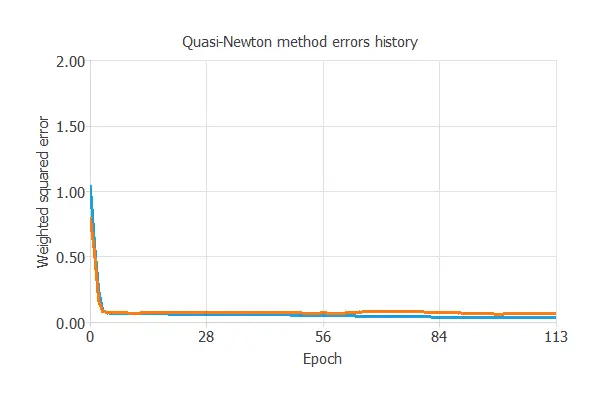
The following chart shows how errors decrease with the iterations during training. The final training and selection errors are training error = 0.054 WSE and selection error = 0.072 WSE, respectively.
5. Model selection
The objective of model selection is to find the network architecture with the best generalization properties, which minimizes the error on the selected instances of the data set.
More specifically, we want to find a neural network with a selection error of less than 0.072 WSE, the value we have achieved so far.
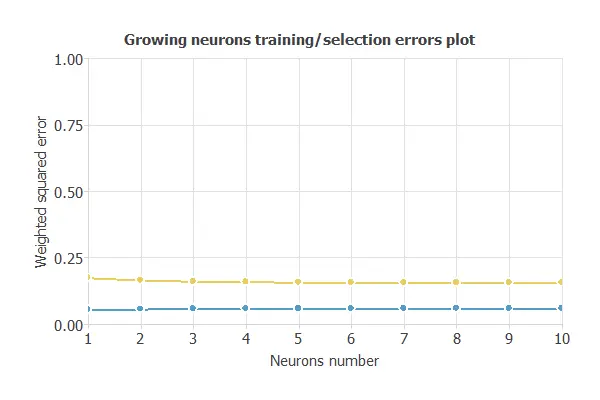
Order selection algorithms train several network architectures with a different number of neurons and select that with the smallest selection error.
The incremental order method starts with a few neurons and increases the complexity at each iteration. The following chart shows the training error (blue) and the selection error (orange) as a function of the number of neurons.
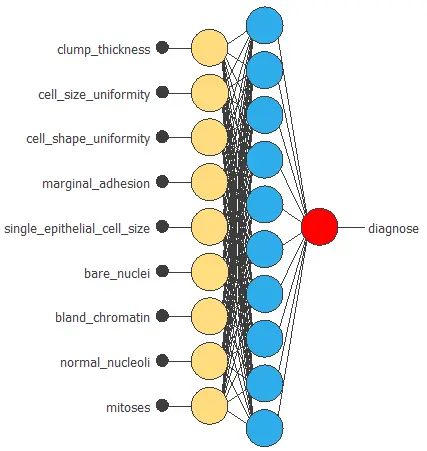
The figure below shows the final architecture for the neural network.
6. Testing analysis
The objective of the testing analysis is to validate the generalization performance of the trained neural network.
To validate a classification technique, we need to compare the values provided by this technique to the observed values. We can use the ROC curve as it is the standard testing method for binary classification projects.
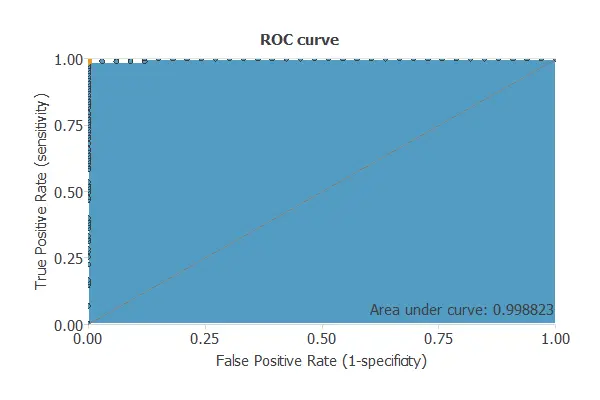
A random classifier has an area under a curve of 0.5, while a perfect classifier has an area under a curve of 1.
In practice, this measure should take a value between 0.5 and 1. The closer to 1, the better the classifier. In this example, this parameter is AUC = 0.804, which means a great performance.
The following table contains the elements of the confusion matrix. This matrix contains the true positives, false positives, false negatives, and true negatives for the variable diagnose.
| Predicted negative | Predicted positive | |
|---|---|---|
| Real negative | 101 | 2 |
| Real positive | 0 | 33 |
The binary classification tests are parameters for measuring the performance of a classification problem with two classes:
- Classification accuracy (ratio of instances correctly classified): 98.5%
- Error rate (ratio of instances misclassified): 1.5%
- Specificity (ratio of real positives that the model predicts as positives): 98%
- Sensitivity (ratio of real negatives that the model predicts as negatives): 100%
7. Model deployment
Once we have tested the neural network’s generalization performance, we can save the neural network for future use in the so-called model deployment mode.
We can diagnose new patients by calculating the neural network outputs. For that, we need to know the input variables for them. An example is the following:
- clump_thickness (1-10): 4
- cell_size_uniformity (1-10): 3
- cell_shape_uniformity (1-10): 3
- marginal_adhesion (1-10): 2
- single_epithelial_cell_size (1-10): 3
- bare_nuclei (1-10): 4
- bland_chromatin (1-10):3
- normal_nucleoli (1-10): 2
- mitoses (1-10): 1
- diagnose: Benignant
The mathematical expression represented by the neural network is written below. It takes the inputs clump_thickness, cell_size_uniformity, cell_shape_uniformity, marginal_adhesion, single_epithelial_cell_size, bare_nuclei, bland_chromatin, normal_nucleoli and mitoses to produce the output diagnose. The information is propagated feed-forward for classification problems through the scaling, perceptron, and probabilistic layers.
scaled_clump_thickness = (clump_thickness-4.442170143)/2.818700075;
scaled_cell_size_uniformity = (cell_size_uniformity-3.150810003)/3.062900066;
scaled_cell_shape_uniformity = (cell_shape_uniformity-3.215229988)/2.986390114;
scaled_marginal_adhesion = (marginal_adhesion-2.830159903)/2.862469912;
scaled_single_epithelial_cell_size = (single_epithelial_cell_size-3.234260082)/2.221460104;
scaled_bare_nuclei = (bare_nuclei-3.544660091)/3.641190052;
scaled_bland_chromatin = (bland_chromatin-3.445100069)/2.447900057;
scaled_normal_nucleoli = (normal_nucleoli-2.869689941)/3.050430059;
scaled_mitoses = (mitoses-1.603219986)/1.731410027; perceptron_layer_1_output_0 = tanh( 0.0344199 + (scaled_clump_thickness*0.181135) + (scaled_cell_size_uniformity*0.217282) + (scaled_cell_shape_uniformity*0.196496) + (scaled_marginal_adhesion*0.125525) + (scaled_single_epithelial_cell_size*-0.0287599) + (scaled_bare_nuclei*0.314836) + (scaled_bland_chromatin*0.0702443) + (scaled_normal_nucleoli*0.18966) + (scaled_mitoses*0.185792) );perceptron_layer_1_output_1 = tanh( -0.0365313 + (scaled_clump_thickness*-0.185626) + (scaled_cell_size_uniformity*-0.224576) + (scaled_cell_shape_uniformity*-0.203225) + (scaled_marginal_adhesion*-0.127989) + (scaled_single_epithelial_cell_size*0.029879) + (scaled_bare_nuclei*-0.321838) + (scaled_bland_chromatin*-0.0715547) + (scaled_normal_nucleoli*-0.194488) + (scaled_mitoses*-0.191226) );perceptron_layer_1_output_2 = tanh( -0.0362404 + (scaled_clump_thickness*-0.184876) + (scaled_cell_size_uniformity*-0.223219) + (scaled_cell_shape_uniformity*-0.202266) + (scaled_marginal_adhesion*-0.127906) + (scaled_single_epithelial_cell_size*0.0295682) + (scaled_bare_nuclei*-0.321034) + (scaled_bland_chromatin*-0.0711169) + (scaled_normal_nucleoli*-0.193701) + (scaled_mitoses*-0.190348) );perceptron_layer_1_output_3 = tanh( -0.0361874 + (scaled_clump_thickness*-0.18509) + (scaled_cell_size_uniformity*-0.223665) + (scaled_cell_shape_uniformity*-0.202517) + (scaled_marginal_adhesion*-0.127643) + (scaled_single_epithelial_cell_size*0.0299014) + (scaled_bare_nuclei*-0.320942) + (scaled_bland_chromatin*-0.0715655) + (scaled_normal_nucleoli*-0.194093) + (scaled_mitoses*-0.190596) );perceptron_layer_1_output_4 = tanh( 0.0360132 + (scaled_clump_thickness*0.184484) + (scaled_cell_size_uniformity*0.222643) + (scaled_cell_shape_uniformity*0.201691) + (scaled_marginal_adhesion*0.12739) + (scaled_single_epithelial_cell_size*-0.0299674) + (scaled_bare_nuclei*0.320025) + (scaled_bland_chromatin*0.0714402) + (scaled_normal_nucleoli*0.193439) + (scaled_mitoses*0.189759) ); perceptron_layer_1_output_5 = tanh( 0.0354705 + (scaled_clump_thickness*0.183133) + (scaled_cell_size_uniformity*0.2203) + (scaled_cell_shape_uniformity*0.199823) + (scaled_marginal_adhesion*0.126653) + (scaled_single_epithelial_cell_size*-0.0295373) + (scaled_bare_nuclei*0.317894) + (scaled_bland_chromatin*0.0708924) + (scaled_normal_nucleoli*0.191821) + (scaled_mitoses*0.188106) ); perceptron_layer_1_output_6 = tanh( 0.0348187 + (scaled_clump_thickness*0.181724) + (scaled_cell_size_uniformity*0.218308) + (scaled_cell_shape_uniformity*0.197597) + (scaled_marginal_adhesion*0.125867) + (scaled_single_epithelial_cell_size*-0.0290229) + (scaled_bare_nuclei*0.315884) + (scaled_bland_chromatin*0.0705261) + (scaled_normal_nucleoli*0.19041) + (scaled_mitoses*0.186599) ); perceptron_layer_1_output_7 = tanh( -0.0355217 + (scaled_clump_thickness*-0.183151) + (scaled_cell_size_uniformity*-0.220295) + (scaled_cell_shape_uniformity*-0.199629) + (scaled_marginal_adhesion*-0.126905) + (scaled_single_epithelial_cell_size*0.0293504) + (scaled_bare_nuclei*-0.318226) + (scaled_bland_chromatin*-0.070681) + (scaled_normal_nucleoli*-0.19183) + (scaled_mitoses*-0.188182) ); perceptron_layer_1_output_8 = tanh( 0.0349867 + (scaled_clump_thickness*0.18201) + (scaled_cell_size_uniformity*0.21809) + (scaled_cell_shape_uniformity*0.197321) + (scaled_marginal_adhesion*0.126417) + (scaled_single_epithelial_cell_size*-0.029256) + (scaled_bare_nuclei*0.316366) + (scaled_bland_chromatin*0.0703615) + (scaled_normal_nucleoli*0.190555) + (scaled_mitoses*0.186607) ); perceptron_layer_1_output_9 = tanh( 0.0344097 + (scaled_clump_thickness*0.181045) + (scaled_cell_size_uniformity*0.217696) + (scaled_cell_shape_uniformity*0.196321) + (scaled_marginal_adhesion*0.125367) + (scaled_single_epithelial_cell_size*-0.0283902) + (scaled_bare_nuclei*0.314796) + (scaled_bland_chromatin*0.0700932) + (scaled_normal_nucleoli*0.189455) + (scaled_mitoses*0.185805) ); probabilistic_layer_combinations_0 = -0.0355815 +0.58858*perceptron_layer_1_output_0 -0.605611*perceptron_layer_1_output_1 -0.603077*perceptron_layer_1_output_2 -0.603653*perceptron_layer_1_output_3 +0.601335*perceptron_layer_1_output_4 +0.596165*perceptron_layer_1_output_5 +0.591153*perceptron_layer_1_output_6 -0.596392*perceptron_layer_1_output_7 +0.59155*perceptron_layer_1_output_8 +0.588449*perceptron_layer_1_output_9 diagnose = 1.0/(1.0 + exp(-probabilistic_layer_combinations_0);You can export the above expression to a dedicated diagnosis software doctors use.
8. Tutorial video
You can watch the step-by-step tutorial video below to help you complete this Machine Learning example for free using the easy-to-use machine learning software Neural Designer.
References
- We have obtained the data for this problem from the UCI Machine Learning Repository.
- Wolberg, W.H., & Mangasarian, O.L. (1990). Multisurface method of pattern separation for medical diagnosis applied to breast cytology. In Proceedings of the National Academy of Sciences, 87, 9193–9196.
- Zhang, J. (1992). Selecting typical instances in instance-based learning. In Proceedings of the Ninth International Machine Learning Conference (pp. 470–479). Aberdeen, Scotland: Morgan Kaufmann.